COVID-19 Primer
UNDERSTANDING THE OPPORTUNITY
The volume of news articles, research, and in some instance’s Presidential briefings recommending new tests, drugs, and other forms of treatment can make finding meaningful opportunities difficult. We believe that understanding the fundamentals of the virus will better enable us to judge the viability of treatments and services that our healthcare system will rely on.
UNDERSTANDING COVID-19
While the news and investors focus on infection rates and the various efficacy of non-pharmaceutical interventions (NPIs) like social distancing, we think it is equally worthwhile to focus on understanding the virus itself. Where does COVID-19 come from, how does it spread, what kind of treatments are being developed, how do we test for it, and lastly, how do we approach COVID-19 in a post-pandemic world.
ORIGIN STORY
The human coronavirus SARS-CoV-2 (COVID-19) is a form of severe acute respiratory syndrome from the same family of viruses as SARS (2003) and MERS (2012). These viruses are highly transferable to humans and livestock, and it’s theorized the initial transmission from bats to humans for SARS and COVID-19 occurred in China. MERS was transferred mainly via camel, but it’s thought that bats act as the MERS reservoir or initial host, originally transferring it to camels.
A long-term study of bats in southern China revealed that 10% of bats tested positive for SARS-related coronavirus nucleotides (RNA)[1]. The presence of virus RNA simply indicates they carry the virus; it doesn’t necessarily mean the bats suffer from it. While many types of coronaviruses exist, only some are transferrable to humans and livestock, particularly those that bond with the human entry receptor ACE2 (angiotensin-converting-enzyme-II). The potential of SARS and SARS-related viruses spreading in hotspots like southern China has been widely studied since SARS in 2003, and “warnings” of additional outbreaks have existed just as long.
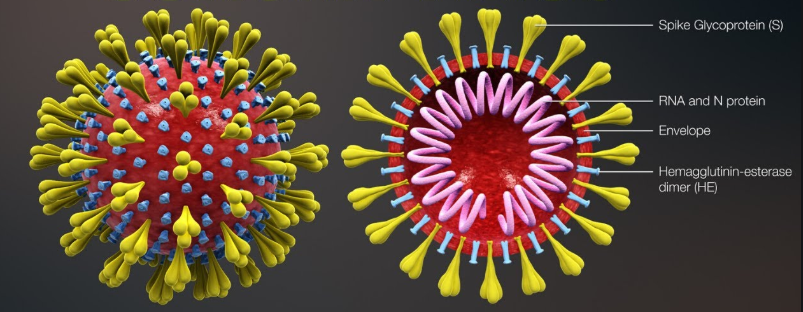
COVID-19 cells are shown In Figure 1 above along with its individual components. The ones we’re interested in are the RNA (ribonucleic acid) and Spike Glycoprotein (Spike). RNA is the means by which the virus will replicate, and the Spike is the mechanism the virus uses to enter a cell. Most research into treatments focus on preventing the virus from being able to enter a host cell; in COVID-19, this would be done by preventing the Spike from bonding with receptor ACE2. This is the same receptor SARS used to enter host cells.
Interestingly, the virus envelope is made of fat lipids which are lysed when they encounter soap. Soap is more effective than alcohol at sterilizing the virus because a small amount of soap can easily cover a large surface area, whereas the virus needs to be submerged in rubbing alcohol to be properly inactivated.
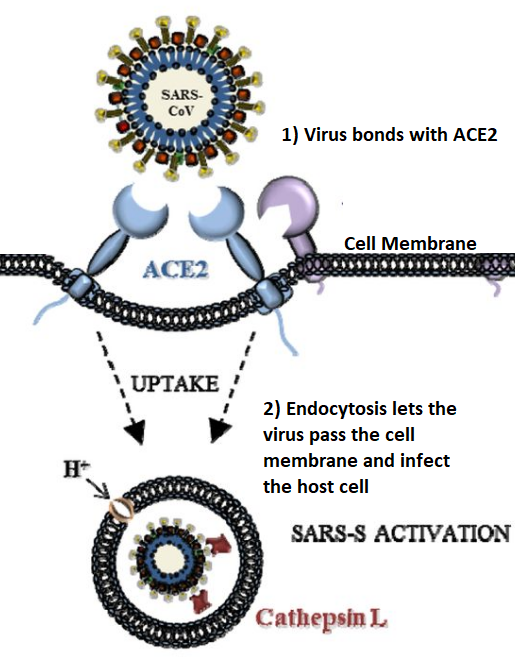
Figure 2 below shows the SARS virus using its Spike to bond with ACE2, and then through endocytosis it passes through the cell membrane. From there the virus can release its RNA into the host cell’s cytoplasm and begin to replicate.
All coronaviruses are “positive-sense single-stranded RNA viruses”, which means they’re capable of replication without using the host cell’s DNA. This means the virus can replicate much faster than ones that rely on DNA, but it also means there is no “back-up copy” of the virus should replication be disrupted or mutated.
Once in the cell the virus releases its RNA into the cytoplasm whereby it instructs ribosomes to form proteins that will build all the components to make another virus (called a “virion”), which will eventually be ejected from the cell to go find another host cell.
DEVELOPING A TREATMENT
There are really two fundamental approaches for reducing the spread and impact of coronavirus, and the best solution is likely a combination of both – through non-pharmaceutical interventions (NPI’s) and pharmaceutical interventions. In this report we will focus on pharmaceutical treatment. Current research is on preventing the virus from entering the host cell (i.e. inhibit the ACE2 receptor from bonding with the virus Spike) or by stopping the virus from replicating once it’s in the host cell. Some examples of existing drugs that use these methods on different viruses are Ribavirin (Hepatitis C) and Lopinavir/ritonavir (HIV/AIDS). Trials are also focusing on anti-inflammatory products (immune suppressant, like Hydroxy chloroquine) as anecdotal observations have indicated it may slow or reduce the severity of the virus. To be sure, while we read optimistically into potential treatments, we also recognize these trial drugs effects on the COVID-19 are not completely understood, and something that seems to help in some patients may make things worse in others. To understand these treatments better, we’ll look at ACE2 and then the process by which a virus RNA replicates.
ACE2 is a protein/enzyme that acts as part of a cells membrane and protrudes in order to bond with other molecules. ACE2’s purpose is to regulate blood pressure by binding and cleaving a specific peptide (amino acid chain) that causes vasoconstriction (narrows arteries). The newly cleaved peptide instead acts as a vasodilator (enlarges arteries) and reduces blood pressure. It can be found in cells of the lungs, arteries, heart, kidney, and intestines, with its highest concentrations
found in the epithelia (exterior lining) of the lung and small intestine. These high concentrations account for the majority of reported symptoms affecting the lungs, nose, throat, and gastrointestinal system. As the virus bonds to more and more ACE2 receptors, the body has less ways to fight vasoconstriction, and so damage to the organs and inflammation can occur.
Readily available blood pressure medicine (antihypertensive drugs) could help slow the spread of COVID-19 between cells by reducing blood pressure, easing inflammation and causing the body to free-up roaming ACE2 enzymes. The ACE2 enzymes initially would not be bound to a cell membrane and therefore could bind the virus without a host cell for the virus to infect, essentially neutralizing the virus. Regulatory approvals for clinical trials involving synthetic ACE2 have been greenlit. One worry is that this treatment could increase the amount of ACE2 receptors on a healthy cell membrane and increase the ability for the virus to spread more than it impedes.
Nitric Oxide (NO) is another potential treatment – it again interacts with ACE2 to encourage vasodilation of cells (reduce blood pressure) and its bioavailability can be easily controlled with oral supplements. NO was also used in the treatment of SARS[1] in 2003 and research into the inhibition of the virus’s replication was proven but the mechanism for inhibition was unknown at the time. A 2009 study determined that NO was interfering with SARS virus’s Spike protein as it tried to bond with ACE2 – this is the same mechanism that COVID-19 uses. Institutions in Germany and the US as well as the University of British Columbia are running trials.
The second method of treating COVID-19 is through stopping the RNA from replicating once it’s in the host cell. In the process of replication (at least for a virus in the corona family) the single RNA strand is read via translation (ribosome “reads” the RNA) and the ribosome builds whatever protein structures the RNA has told it to build – i.e. the other components of the virus, including more RNA. The newly created virus leaves the host cell via exocytosis in order to seek other host cells and this process is repeated until the host cell dies or explodes, causing an immune-response and inflammation, as well as shedding (when cells are ejected from the epithelial – shedding in the lungs can cause mucus, puss, inflammation, and more).
Treatments that target RNA replication seek to interrupt RNA translation (“reading”) that the ribosome does. This is done mainly through two approaches. The first approach is to have the ribosome build the new RNA (or other protein) incorrectly, so the virus can’t replicate its components properly, and the second approach is to inhibit the capping mechanism on newly created RNA (specifically mRNA – messenger RNA). RNA without 5’ and 3’ caps on either end of its genome that are located outside of the nuclease of a cell are destroyed in the cytoplasm.
Even considering the current superior understanding of the corona family viruses and its interactions with human cells compared to our knowledge during the SARS pandemic in 2003, we still do not have a clear method to treat COVID-19.
TESTING
With the rapid spread of COVID-19 around the world (one person generally infecting 2-3 others), proper testing and diagnosis of COVID-19 is absolutely crucial for shaping government action and resource allocation. As of April 7, 2020, there are 109 diagnostic tests available for use[2] – with most being approved via “emergency use authorization” from the companies corresponding government health organization. Of those 109 diagnostic tests, 62 are PCR (Polymerase Chain Reaction), 43 are Serological, and the remaining use various other methods.
The most common test, rRT-PCR (reverse transcription polymerase chain reaction), is offered by the majority of the players in the testing kit and medical device landscape including Abbott and Thermo-Fisher. Most of these testing kits have been Emergency Use Authorized (EUA)[3], and so have not been approved through regular FDA channels. In order to conduct a PCR test, a nasal swab is used to gather a sample (see figure 3 below – the sample is typically taken between the upper throat and nose). In case of shortage of nasal swabs (from suppliers like Becton Dickinson, Copan Diagnostics, Puritan Medical Products, and Thermo-Fisher), less effective throat swabs are used in lieu.
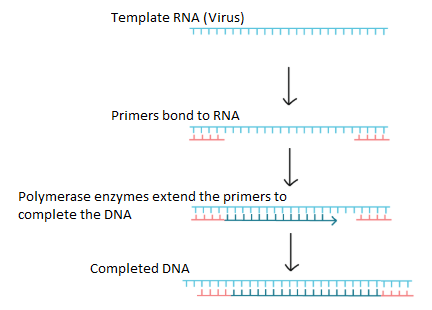
RT-PCR involves turning virus RNA into DNA (“reverse transcription”) and then multiplying the resulting DNA using polymerase chain reaction. Each rRT-PCR testing kit will contain: reverse transcriptase (encourages RNA bonding to nucleotides), dNTPs (nucleotides: Adenine, Guanine, Cytosine, Uracil, Thymine), primers (initially bond to RNA and encourages DNA synthesis), template RNA (inactive virus), and necessary enzymes like Taq polymerase which makes new strands of DNA using existing strands as templates. Figure 4 below shows the process of reverse transcription.
Once the DNA is completed via reverse transcription, the DNA undergoes polymerase chain reaction. The DNA is heated up and denatured into its two RNA components. The two RNA strands are cooled down, and the entire process is then repeated. The two RNA will become two DNA strands, which are then denatured into four RNA, etc. until a certain length or mass of DNA has been synthesized. This typically can take between 2 to 4 hours and is considered a highly complex procedure.
Fluorescent binders or probes are used in the solution and are activated in various ways, but all are excited due to the process of RNA being synthesized into DNA. As such, the determination of the presence of the virus is directly related to the amount of light given off by the rRT-PCR test. If virus RNA isn’t present in the test sample, then no DNA will be synthesized, and no illumination will occur.
In the US, the CDC’s Clinical Laboratory Improvement Amendments (CLIA) determines what types of testing, calibration, and labs can process test kits. CLIA ranks procedures as “waived”, “moderately complex”, or “highly complex”. Waived indicates a procedure has been reviewed, and with simple training and apparatus the test is repeatable with a very low margin for error, and thus if the manufacturers recommendations are followed the lab needs no certificate to perform the test. Nonwaived testing means the CLIA needs to have inspected the lab and approved the labs quality system standards (proficiency testing, quality control and assessment, and personnel requirements). Complex waived and non-waived tests are recommended to be calibrated via proficiency testing, which is what all rRT-PCR tests fall under. Typical controls for testing include a negative control (no RNA), and a positive template (should test positive for COVID-19), which need to be run for each test in some cases.[1]
A BIT OF QUARANTINE EARLY GOES A LONG WAY
With the US economy struggling due to mandated business closures, social distancing, and self quarantine, some have started to wonder if the “cure is worse than the disease.” Are these measures worth the stress the average individual is under? To understand the logic at the base level for the government quarantine mandates, we look no further than Iceland.[2]
Iceland identified its first case of COVID-19 at the end of February and immediately implemented a screening process where individuals who appeared symptomatic, had recently traveled to high-risk countries, or were in contact with someone infected were tested for the virus. Of these targeted tests, a whopping 13.3% tested positive. Anyone confirmed positive was subject to a two-week quarantine. In March, while targeting these high-risk individuals, Iceland also asked for low-risk individuals to be tested– it was a random sample of the population and Iceland could compare the infection rate of high-risk individuals to those with low risk. Of those considered low-risk, 0.8% tested positive. Over the next twenty days that 0.8% remained stable. Unlike other countries that didn’t respond as quickly, Iceland saw no increase in the rate of the spread of the virus in their general population. They had already flattened the curve before it could begin to ramp up.
What do we take away from this? Iceland proves their immediate response of targeted testing and quarantine methods worked, and the spread of the virus can be slowed. Of course, Iceland is not like most countries. Its population is relatively small and highly concentrated around the capital (60%), making monitoring and contact tracing very straightforward. Places like the US and Europe would have a much more difficult time and would need massive scale to be anywhere near as effective as Iceland. Fundamentally, quarantining works. The question going forward is can countries like the US ever have the political will to implement such an aggressive and widespread quarantine policy early-on during future pandemics in order to stop the spread, or are we resigned to always chase the “flattening curve?”
POST-PANDEMIC
Unfortunately, looking to the past to help provide clarity for the future is unsettling in these types of situations. The consolidation and leverage of knowledge from previous pandemics, especially SARS in 2003, aren’t at a scale for which researchers and governments can make confident assumptions in the treatments for COVID-19. The medical system was unprepared for the loads it saw during SARS and it is again unprepared for COVID-19, when most subject matter experts agree it was a matter of “when” and not “if” for another pandemic. Part of the issue is that research into treatments of SARS (2003) was limited due to access to infected patients, governments various priorities and responses, the lack of a uniform governing body to help organize and collect research information, as well as the reporting of anecdotal treatments that may or may not actually work[3]. These are all issues we see being dealt with again, today.
Testing and validation of testing of COVID-19 will remain a large focus until a herd immunity is established, which will be driven by a vaccine. With a vaccine target of 8-12 months, it appears next flu season is when people will be inoculated against COVID-19 and testing can be reduced to severe cases. Like SARS and other vaccinated diseases, COVID-19 will be a reality we will likely have to live with like the common cold.
PATHFINDER ACTIONS
With all that said, this is where we branch into a company of interest to Pathfinder – Microbix Biosystems (TSX:MBX). Microbix is a Canadian company that develops and sells biological products and technologies to distributors and labs. Microbix leads in the supply of antigens, which are proteins that interact with antibodies. Typical use of antigens is to simulate viruses, such as HIV or HPV, and these synthetic virus antigens are then used in labs to test the quality of the lab’s procedures. As testing for viruses becomes more automated and more efficient, it becomes paramount that the calibration of the lab equipment is correct. A hundred swabs of COVID-19 tests run using a machine that was incorrectly reading false positives (identifies COVID when the sample actually has none) or false negatives (identifies no COVID-19 when it is actually present) would be detrimental to the labs schedule (re-testing takes days), resources (order new swabs, new test kits), and most importantly the patients treatment. In a worst-case scenario, you could have false negatives re-entering society while still infectious and increasing the spread of the virus. Enter Microbix, which has developed a COVID-19 Quality Assessment Product (QAP) that allow labs to calibrate their testing equipment so that labs can be confident that their equipment is providing the correct results.
The World Health Organization (WHO), Federal Drug Administration (FDA), and Health Canada all mandate labs have quality assurance programs and recommend the use of external quality assessment products. We anticipate greater growth will occur in the companies that develop test kits, swabs, as well as quality assurance products that are directly related to COVID-19. While Microbix has other lines of QAP and other medical equipment, we see their COVID-19 offering as a major catalyst for the near future earnings.
APPENDIX I
|
[1] “Bat Coronaviruses in China”, Yi Fan, Kai Zhao, Zheng-Li Shi, and Peng Zhou, MDPI, 2019
[2] “Dual effect of nitric oxide on SARS-CoV replication: viral RNA production and palmitoylation of the S protein are affected”, Virology, 2009, ncbi.nlm.nih.gov/pubmed/19800091
[3] “Coronavirus Test Tracker: Commercially Available COVID-19 Diagnostic Tests”, https://www.360dx.com/coronavirus-test-tracker-launched-covid-19-tests
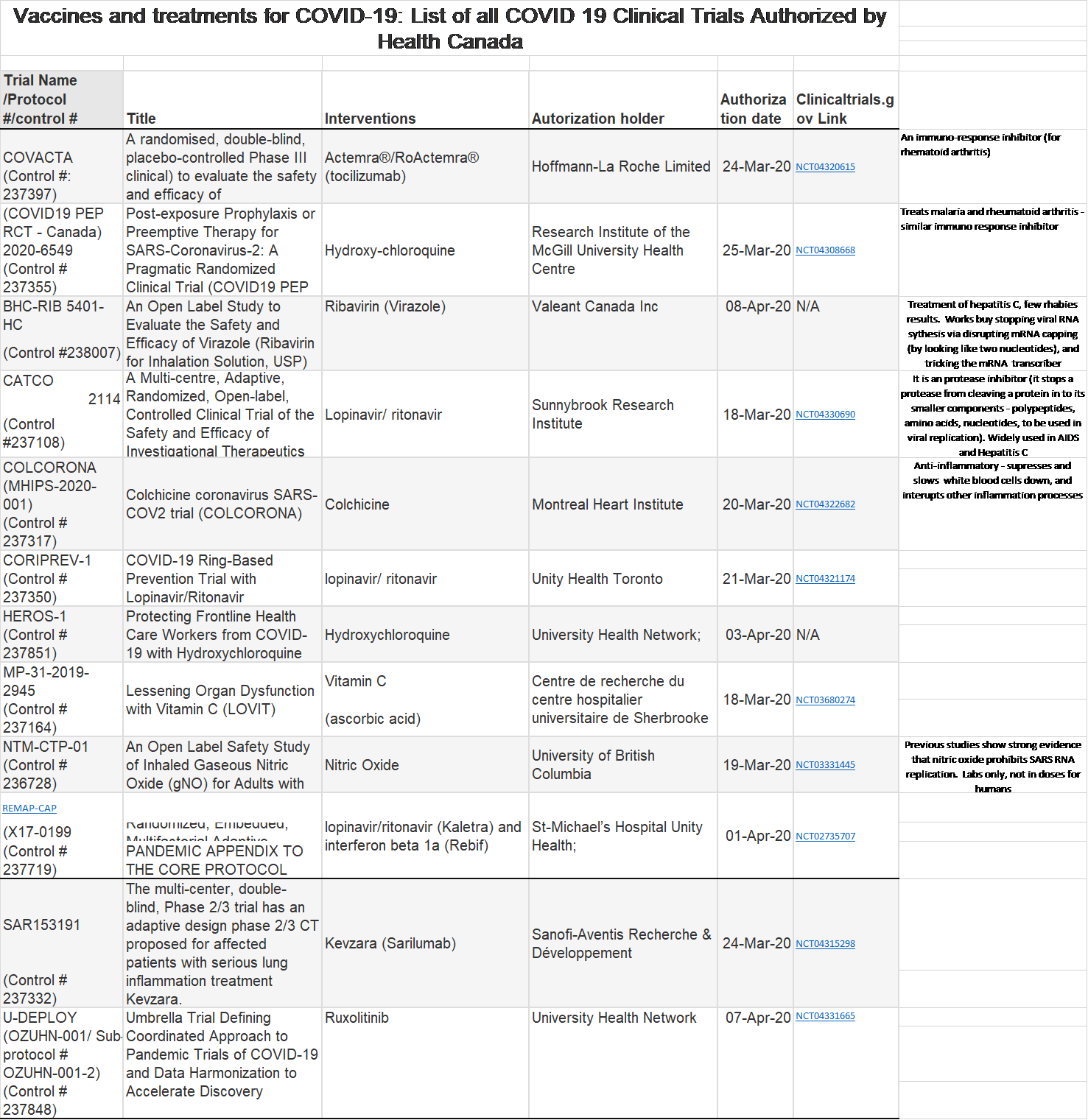
[4] https://www.visualcapitalist.com/every-vaccine-treatment-covid-19-so-far/
[5] “Accelerated Emergency Use Authorization (EUA) Summary COVID-19 RT-PCR Test (Laboratory Corporation of America)”, https://www.fda.gov/media/136151/download
[6] “Spread of SARS-CoV-2 in the Icelandic Population” Daniel F. Gudbjartsson, Ph.D., Agnar Helgason, Ph.D., Hakon Jonsson, Ph.D., Olafur T. Magnusson, Ph.D., Pall Melsted, Ph.D., Gudmundur L. Norddahl, Ph.D., Jona Saemundsdottir, B.Sc., Asgeir Sigurdsson, B.Sc., Patrick Sulem, M.D., Arna B. Agustsdottir, M.Sc., Berglind Eiriksdottir, Run Fridriksdottir, M.Sc, April 14, 2020
[7] Institute of Medicine (US) Forum on Microbial Threats; Knobler S, Mahmoud A, Lemon S, et al., editors. Learning from SARS: Preparing for the Next Disease Outbreak: Workshop Summary. Washington (DC): National Academies Press (US); 2004. LESSONS FROM SARS FOR FUTURE OUTBREAKS. Available from: https://www.ncbi.nlm.nih.gov/books/NBK92465/
National Instrument 31-103 requires registered firms to disclose information that a reasonable investor would expect to know, including any material conflicts with the firm or its representatives. Doug Johnson and/or Pathfinder Asset Management Limited are an insider of companies periodically mentioned in this report. Please visit www.paml.ca for full disclosures.
*All returns are time weighted and net of investment management fees. Returns from the Pathfinder Partners’ Fund and Pathfinder Real Fund are presented based on the masters series of each fund. The Pathfinder North American Equity Portfolio and The Pathfinder North American Income Portfolio are live accounts. These are actual accounts owned by the Pathfinder Chairman (Equity) and client (High Income) which contain no legacy positions, cash flows or other Pathfinder investment mandates or products. Monthly inception dates for each fund and portfolio are as follows: Pathfinder North American Equity Portfolio (January 2011), Pathfinder North American High-Income Portfolio (October 2012) Pathfinder Partners’ Fund (April 2011), Pathfinder Real Fund (April, 2013), and Pathfinder International Fund (November 2014).
Pathfinder Asset Management Limited (PAML) and its affiliates may collectively beneficially own in excess of 10% of one or more classes of the issued and outstanding equity securities mentioned in this newsletter. This publication is intended only to convey information. It is not to be construed as an investment guide or as an offer or solicitation of an offer to buy or sell any of the securities mentioned in it. The author has taken all usual and reasonable precautions to determine that the information contained in this publication has been obtained from sources believed to be reliable and that the procedures used to summarize and analyze such information are based on approved practices and principles in the investment industry. However, the market forces underlying investment value are subject to sudden and dramatic changes and data availability varies from one moment to the next. Consequently, neither the author nor PAML can make any warranty as to the accuracy or completeness of information, analysis or views contained in this publication or their usefulness or suitability in any particular circumstance. You should not undertake any investment or portfolio assessment or other transaction on the basis of this publication, but should first consult your portfolio manager, who can assess all relevant particulars of any proposed investment or transaction. PAML and the author accept no liability of any kind whatsoever or any damages or losses incurred by you as a result of reliance upon or use of this publication.